RESEARCH
In vitro Metabolic Engineering
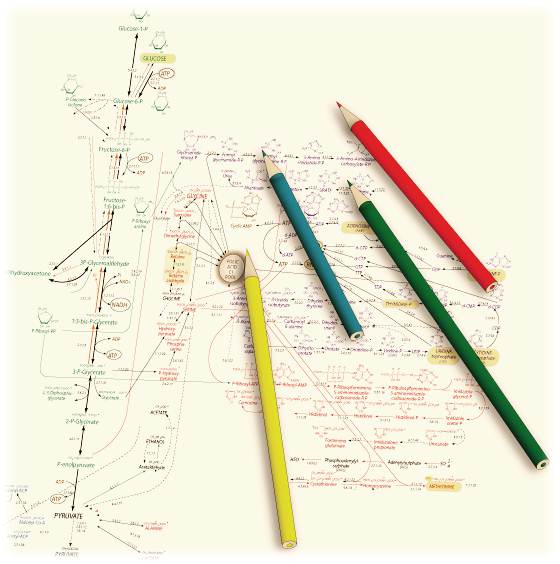 Biocatalytic production of value-added chemicals from renewable feedstocks
has been widely recognized as a key technology to promote sustainability
of chemical industries. The best known among them is production of biofuels,
represented by bioethanol and biodiesel. Demands for biofuel have been
increasing globally and intensive studies have been done for the development
of novel production processes that enable “faster” and “more abundant”
production. On the other hand, it is imaginable that production of more
variety of chemical compounds, including polymers, pigments, food additives,
and pharmaceuticals, will be the next challenge in this field. To achieve
this, development of a “simpler” and “more universal” biocatalytic production
system will be necessary.
Biocatalytic production of value-added chemicals from renewable feedstocks
has been widely recognized as a key technology to promote sustainability
of chemical industries. The best known among them is production of biofuels,
represented by bioethanol and biodiesel. Demands for biofuel have been
increasing globally and intensive studies have been done for the development
of novel production processes that enable “faster” and “more abundant”
production. On the other hand, it is imaginable that production of more
variety of chemical compounds, including polymers, pigments, food additives,
and pharmaceuticals, will be the next challenge in this field. To achieve
this, development of a “simpler” and “more universal” biocatalytic production
system will be necessary.
Thermophilic enzymes are getting increased use in biocatalytic production
processes. Their excellent stability and high reaction temperature have
advantages, such as better substrate solubility, a high mass transfer rate,
and the low risk of contamination. Recombinant DNA techniques have allowed
the heterologous expression of thermophilic enzymes in mesophilic organisms
(e.g. Escherichia coli). Use of recombinant mesophiles having thermophilic
enzymes at high temperature results in denaturation of indigenous enzymes
and elimination of undesired side reactions. This approach is, in principle,
available for all thermophilic enzymes only if they can be functionally
expressed in mesophilic hosts. Combination of multiple thermophilic enzymes
allows us to construct a bespoke metabolic pathway in vitro. Since the
reaction proceeds independently of the metabolic status of host cells,
process controls can be thoroughly simplified and the production yield
is thermodynamically predictable. We referred to this strategy as “Invitro
metabolic engineering (SME)” and are now in attempt to construct pathways
for the production of a variety of chemical compounds.
Nonaqueous Bioprocess
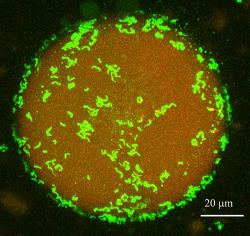
Confocal micrograph of a water-in-oil emulsion stabilized by a hydrophobic
bacterium, Rhodococcus opacus.
R. opacus cells (green) are preferably localized at a interfacial area
between aqueous (red background) and organic-solvent (dark background)
phases
(Honda K et al, Appl Microbiol Biotechnol 2008)
The use of organic solvents as reaction media has the potential to greatly
expand the repertoire of bioprocess. Successful examples of biocatalytic
conversions in nonaqueous media using enzymes including lyophilized lipases,
esterases, and proteases have been reported. However, most of these biocatalytic
conversions involve relatively simple, single-step hydrolytic enzymes.
One of the challenges in this area is the utilization of more complex enzymatic
reactions involving multicomponent enzymes, cofactor regeneration, and
multistep enzymatic conversions in organic solvent. Therefore, attention
has been paid to the development of a whole-cell catalyst that functions
in nonaqueous environment.
Rhodococcus opacus B-4 cells exhibit a high affinity for water-immiscible
hydrocarbons and are dispersible in essentially water-free organic solvents.
Due to these hydrophobic features, R. opacus B-4 can easily access to the
water-immiscible chemicals dissolved in organic solvnets and have an advantage
in taking up them. By using R. opacus B-4 cells as “hydrophobic biocatalytic
particles”, we are developing nonaquesous bioprocesses for the production
of water-immiscible chemicals.
How can we fully realize P recycling on a large scale?
Increased attention has been paid to P recovery and recycling for sustainable P management. However, P recycling has never been realized on a large scale, mainly because of economical reasons. Obviously, how we can make P recycling a successful business is likely the most important issue to be considered for fully realizing P recycling. The emerging issues on P recycling are relevant to the quality, quantity, cost, and market of recovered P. To tackle these issues, we have recently developed an innovative technology for P recovery using amorphous calcium silicate hydrates (ACSHs) as inexpensive adsorbents. Conventionally, calcium silicate hydrates (CSHs), which are by-products of the building material industry (tobermorite), have been used to recover P from P-rich liquor by crystallization. Our newly synthesized ACSHs remarkably improved the rate and efficiency of P recovery from anaerobic sludge digestion liquor compared to tobermorite. This finding allowed us to create a new business on the P recovery using ACSHs, based on the business-academia collaboration. In our business model, a cement company manufactures ACSHs at low costs, carries them to a P recovery site (e.g. wastewater treatment plant) using agitating trucks, and takes out ACSHs containing more than 15% P2O5 to a fertilizer company. No expensive P recovery plant needs to be constructed at the recovery site any longer. The ACSHs can be directly used as a by-product phosphate fertilizer by the fertilizer company. Importantly, ACSHs can be synthesized using unlimitedly available resources such as calcium and silicate.To fully realize the P recycling, it is essential to build close collaboration between different industrial sectors. To do this, the P Recycling Promotion Council of Japan (PRPCJ) was established as a nationwide association under the support of four important ministries of Japan in 2008. With the assistance of PRPCJ, P recovery has been extended to a variety of manufacturing industries such as chemical industry, food and fermentation industry, automobile industry, and high-tech industries for the production of computer chips and liquid crystal panels. Japan has no significant reserve of rock phosphate and imports approximately 750 kton P/Y from foreign countries, mainly from China. Interestingly, however, 330 ktonP/Y (approximately 46% of total inflow) is imported in the form of food, feed, coal, and iron ore. There exists a variety of un-used P resources such as sewage sludge, steel-making slag, and food waste in Japan. The total amount of the potential P resources is estimated to be 280 ktonP/Y, whereas the P demand for agricultural production in Japan is approximately 300 ktonP/Y. If a sound strategy is developed and applied, P recycling could supply the P demand from the agricultural production in Japan. In this talk, our current efforts to fully realize P recycling on a national level will be presented as a case study.
(This paper was presented at ETH, Zurich, on 15th July, 2011)
バナースペース
BIOCHEMICAL ENGINEERING LAB
Dept.of Biotechnology
Graduate School of Engineering,
Osaka University
Yamadaoka 2-1, Suita Osaka
565-0871 Japan
Phone:+81-6-6879-7437
Fax :+81-6-6879-7439